LINKED PAPER
Water vapour conductance of passerine nest walls. Deeming D.C., Gilchrist R., Szafraniec M. & Pollins J.M. 2020. Acta Ornithologica. DOI: 10.3161/00016454AO2020.55.1.002. VIEW
Ornithologists really don’t know how bird nests work during incubation and chick rearing. Aside from roles in parasite control or sexual selection (Mainwaring et al. 2014) nests have main role as the site of incubation and chick rearing. In this capacity they provide thermal insulation (Deeming et al., 2020), protection from the elements (Biddle et al. 2019), and support adult and the clutch of eggs and nestlings (Biddle et al. 2018a). Nests exhibit both inter- and intra-specific variation in composition (e.g. Biddle et al. 2018b; Briggs et al. 2019) and proportions of moss, grass and hair are important in explaining variation in thermal and hydrological properties of nests (Biddle et al. 2019; Deeming et al., 2020).
A critical aspect of incubation is the maintenance of humidity within the nest to ensure that there is an appropriate rate of water loss from the egg, which maintains embryonic viability. Water loss from an egg is a function of the eggshell porosity and the humidity in the nest (Deeming, 2011). During the 1970s and 1980s there was extensive research on porosity of eggshells, measured as water vapour conductance (WVC), but there was little attention on the water vapour conductance of the nest wall. Those few studies that considered this aspect assumed that water loss from a nest cup would equal that from the egg and water vapour needed to create humidity in the nest came mainly from the egg (Walsberg, 1980; Rahn et al., 1983). Unfortunately, there are no reports of studies that measured nest wall WVC to check these assumptions, possibly because of the technical difficulties of measuring rates of water loss across the nest wall.
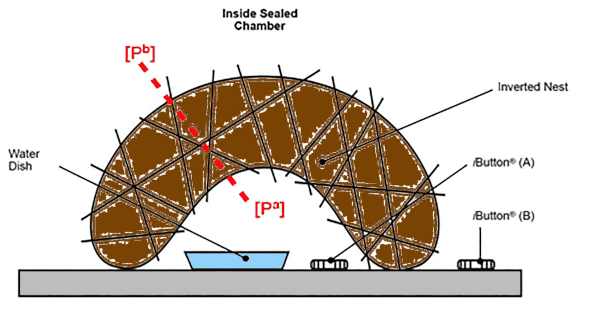
Using small hygrometer-thermometer loggers it was possible to measure the rate of water loss from a water reservoir in the nest cup across a nest wall (Figure 1). Applying Fick’s law (Ar et al., 1974), it was possible to calculate wall WVC for nests built by nine species of European passerine. It was found that the nest wall was very leaky to water vapour (Figure 2). There were differences in WVC values among species, and values for nest wall WVC were two orders of magnitude higher than eggshell WVC values for the same species.
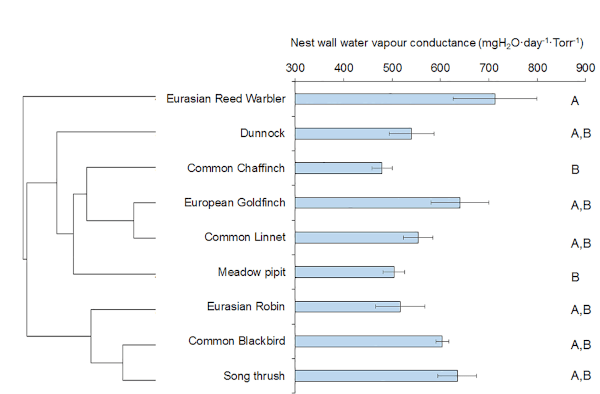
Given nest structure can often be quite open, these results are not perhaps unsurprising, but they have implications for control of nest humidity during incubation.
Previous studies have shown that birds seem incapable of controlling humidity in the nest (e.g. Walsberg, 1983) and the assumption was that the presence of the bird on the nest kept the water vapour leaving the eggs in the nest cup to allow humidity to rise above ambient levels. However, these early studies (Walsberg, 1980; Rahn et al., 1983) were based on a steady-state situation and did not consider the dynamic nature of incubation.
Birds regularly leave the nest to forage or get up to turn the eggs and, when they do, all of the humidity in the nest cup is lost by diffusion to the ambient air. On average passerines can leave their nests twice an hour and can turn their eggs five times an hour (Deeming, 2002), so how does a leaky nest wall affect the build-up of humidity each time they return to incubation?
Here the dynamic nature of intermittent incubation was studied by calculating the water budget for the nest cup over a 12-hour period of intermittent incubation. This was then compared to the amount of water normally lost from a clutch of eggs over the same period. The mass of water lost from the egg was around 700-1,000 mg over 12 hours. However, assuming that water vapour would be lost through the nest wall and when the bird rose from the nest, it was found that to regularly build-up nest cup humidity required 8-9 times more water. The assumption that nest humidity was created by water vapour lost from the eggs accumulating in the cup may work in a steady-state system, but it doesn’t work in a more realistic dynamic incubation environment.
Intermittent incubation of a clutch of eggs in a nest with a wall that easily allows water vapour to diffuse through it requires an additional source of water vapour to raise nest humidity above that of the ambient air. It is known this happens, so where does the extra water come from?
The nest wall may yield water as any plant materials dry out, but this is a limited and unreliable source and unlikely to last long enough over incubation. Walsberg (1980) suggested that evaporation from the skin could be important. Rates of cutaneous water loss are sufficient to allow for humidity build up and are known to be under physiological control. Studies have shown that cutaneous water loss increases during incubation (Piersma & Morrison 1994). It is suggested that the brood patch may not only provide heat energy to raise egg temperature but may be a source of water to quickly raise humidity in the nest cup.
It was concluded that in leaky nests, humidity over time is probably maintained in a dynamic incubation environment by water being lost across the skin but there needs to be further study to confirm this hypothesis.
References
Ar, A., Paganelli, C.V., Reeves, R.B., Greene, D.G. & Rahn, H. 1974. The avian egg: water vapour conductance, shell thickness, and functional pore area. Condor 76: 153–158. VIEW
Biddle, L.E., Broughton, R.E., Goodman, A.M. & Deeming, D.C. 2018b. Composition of bird nests is a species-specific characteristic. Avian Biology Research 11: 132-153. VIEW
Biddle, L.E., Deeming, D.C. & Goodman, A.M. 2018a. Birds use structural properties when selecting materials for different parts of their nests. Journal of Ornithology 159: 999-1008. VIEW
Biddle, L.E., Dickinson, A., Broughton, R.E., Gray, L.A., Bennett, S.L., Goodman, A.M. & Deeming, D.C. 2019. Construction materials affect the hydrological properties of bird nests. Journal of Zoology 309: 161-171. VIEW
Briggs, K.B. Biddle, L.E. & Deeming, D.C. 2019. Geographical location affects size and materials used in the construction of Pied Flycatcher (Ficedula hypoleuca) nests. Avian Research 10: 17. VIEW
Deeming, D.C. 2002. Avian Incubation: Behaviour, Environment and Evolution. Oxford University Press, Oxford.
Deeming, D.C. 2011. Importance of nest type on the regulation of nest humidity in birds. Avian Biology Research 4: 23-31. VIEW
Deeming, D.C., Dickinson, A.M., Broughton, R.E., Locke, E., Gray, L.A., Bennett, S.L., Gilchrist, R., Muniz, S., Goodman, A.M. & Biddle, L.E. 2020. Factors affecting thermal insulation of songbird nests as measured using temperature loggers. Physiological and Biochemical Zoology in press. VIEW
Mainwaring, M.C., Hartley, I.R., Lambrechts, M.M. & Deeming, D.C. 2014. The design and function of birds’ nests. Ecology and Evolution 4: 3909-3928. VIEW
Piersma T. & Morrison R.I.G. 1994. Energy expenditure and water turnover of incubating ruddy turnstones: high costs under high arctic climatic conditions. Auk 111: 366–376. VIEW
Rahn H., Krog J., & Mehlum F. 1983. Microclimate of the nest and egg water loss of the eider Somateria mollissima and other waterfowl in Spitsbergen. Polar Research 1: 171–183. VIEW
Walsberg G. E. 1980. The gaseous microclimate of the avian nest during incubation. American Zoologist 20: 363–372. VIEW
Walsberg G. E. 1983. A test for regulation of nest humidity in two bird species. Physiological Zoology 56: 231–235. VIEW
Image credit
Top right: Typical nest © D. C. Deeming




