
Report from a BOU-funded project
Avian and human visual systems are strikingly different: while we humans have three types of photoreceptors in our retinas that allow us to perceive a varied range of colours, birds have a fourth type that extends such range even further into the ultra-violet (UV) light spectrum. This is why in many studies we use methods to objectively measure and analyse plumage colouration, accounting for perception differences. Reflectance spectrometry allow us to measure the amount of light reflected at different wavelengths by a surface of interest. Plain feather patches can be easily measured with this method, but how do we study plumage patches that combine spots, lines, or even more complicated patterns of dark versus light colours?
One alternative is to make use of digital photography and image-processing techniques to analyze the two-dimensional distribution of the darker/clearer areas that make up a visual pattern (Mason and Bowie 2020). This approach has been used, for example, to analyse Cuttlefish (Sepia spp.) camouflage markings (Barbosa et al. 2008; Chiao et al. 2009), the variation in pigmentation patterns on eggshells (Stoddard and Stevens 2010, Stoddard et al. 2014) and has also been used to analyse plumage patterns (see for example Gluckman & Cardoso 2009, Gluckman & Mundy 2013). This allows us to better understand the possible functions of different types of plumage patterns and the selective forces promoting their evolution. For example, patterns in general are more cryptic against heterogeneous backgrounds, and thus could have a camouflaging function. But regular patterns – such as barred plumages – could also signal plumage condition (and thus may have evolved through sexual selection), as deviations are more salient in a regular pattern (see Gluckman & Cardoso 2009).

Figure 1 Museum skins of Yellow Warblers (Setophaga petechia) kept at the American Museum of Natural History (NY, USA).
My objective with this project was to apply some of these techniques to analyse plumage pattern variation and evolution among a group of closely related passerine birds, the New World warblers of the Setophaga genus (Parulidae). The Setophaga genus now comprises 34 species of the Parulidae family (including all the species that once belonged to the Dendroica genus, see Lovette et al. 2010). While many of these species exhibit patterns of stripes or spots in different regions of their bodies, several others do not, making them an interesting model for to study when and why these patterns may have appeared. Also, in many cases only males exhibit these patterns, indicating sexual selection may have played an important role in their evolution.
I visited the American Museum of Natural History, which houses one of the largest ornithology collections in the world (Figure 1), and I first met with Dr. Mary C. Stoddard, and Audrey Miller, currently a PhD student at Dr. Stoddard´s Lab at Princeton University. They trained me on how to take the photographs I need for the analyses, which was an interesting process. Using a camera and lens specially modified to capture UV light, and a set of filters in a custom-made holder, I took photographs of the specimens in different views. I had to lock myself in a room isolated from any light source, so that the specimen would only reflect the light from a special lamp that emits in all human-visible wavelengths and UV. I would then take one photo with an UV-filter on, and one photo with a visible-light filter on. These photos need to be superimposed afterwards to compose the required image for analyses, so they have to be identical. Therefore, not moving the specimen or the camera was key to the process. And of course, it meant I had to repeat and repeat the process on many occasions…
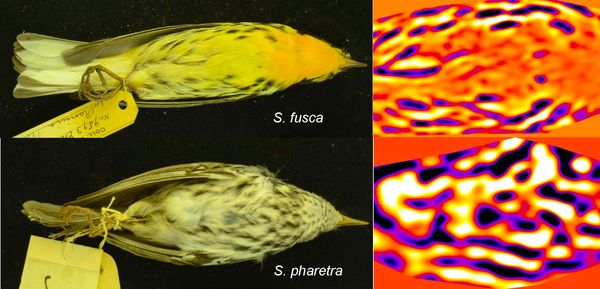
Figure 2 Energy maps generated after applying Fast Fourier bandpass filtering to ventral photographs of a Blackburnian Warbler (Setophaga fusca) and an Arrowhead warbler (S. pharetra).
I obtained photographs of three views (dorsal, ventral, and lateral) of up to five males for all the species in the genus, and I started to analyse these photographs using “granularity” methods, which are based on Fast Fourier bandpass filtering. Briefly, these filters act like sieves of different grain size, and as each filter is applied to the photo, the method assigns an “energy” level based on how well the markings in the photo fit the grain size of each filter. The higher the energy, the better that filter size fits the pattern we are analysing. If a pattern has big and small spots, two different filter sizes can have peak energy values. As an example, in Figure 2 you can see the energy maps generated for the filter sizes with highest energy for ventral views of two warbler species (Blackburnian Warbler Setophaga fusca and the Arrowhead warbler S. pharetra).
These techniques provide numerical variables to describe the different plumage patterning, which I will use to explore the evolution of plumage patterns. As a comprehensive phylogenetic hypothesis for the group is available, phylogenetic comparative methods will be used to determine the origin of different patterned patches, to perform ancestral state reconstruction and to test the effect of habitat characteristics as a selective force. Overall, this study allowed me to greatly expand my knowledge and experience on how to perform colour analyses, and will provide me with a framework for future studies investigating the evolution of complex plumage phenotypes.
Funding
In 2017, Natalia was awarded a BOU Career Development Bursary grant of £2,500 for a project titled “Evolution of plumage patterns in a continental radiation of passerines: the Setophaga warblers” when she was a Postdoc Fellow at the Natural History Museum of Buenos Aires, Argentina.
References
Barbosa, A., Mäthger, L. M., Buresch, K. C., Kelly, J., Chubb, C., Chiao, C. C., & Hanlon, R. T. 2008. Cuttlefish camouflage: the effects of substrate contrast and size in evoking uniform, mottle or disruptive body patterns. Vision Research 48: 1242-1253. VIEW
Chiao, C. C., Chubb, C., Buresch, K., Siemann, L., & Hanlon, R. T. 2009. The scaling effects of substrate texture on camouflage patterning in cuttlefish. Vision Research 49: 1647-1656. VIEW
Cuthill, I. C. 2006. Color perception. In: Hill, G. E. & McGraw, K. J. (eds.) Bird coloration. Volume I, pp. 3-40. Harvard University Press: Cambridge, MA, USA and London, UK.
Gluckman, T. L., & Cardoso, G. C. 2009. A method to quantify the regularity of barred plumage patterns. Behavioral Ecology and Sociobiology 63: 1837-1844. VIEW
Gluckman, T. L., & Mundy, N. I. 2013. Cuckoos in raptors’ clothing: barred plumage illuminates a fundamental principle of Batesian mimicry. Animal Behaviour 86: 1165-1181. VIEW
Lovette, I. J., Pérez-Emán, J. L., Sullivan, J. P., Banks, R. C., Fiorentino, I., Córdoba-Córdoba, S., Echeverry-Galvis, M., Barker, F.K., Burns, K.J. Klicka, J., Lanyon, S.M. & Bermingham, E. 2010. A comprehensive multilocus phylogeny for the wood-warblers and a revised classification of the Parulidae (Aves). Molecular Phylogenetics and Evolution 57: 753-770. VIEW
Mason, N. A., & Bowie, R. C. 2020. Plumage patterns: Ecological functions, evolutionary origins, and advances in quantification. The Auk 137: ukaa060. VIEW
Stoddard, M. C., Kilner, R. M., & Town, C. 2014. Pattern recognition algorithm reveals how birds evolve individual egg pattern signatures. Nature Communications 5. VIEW
Stoddard, M. C., & Prum, R. O. 2011. How colorful are birds? Evolution of the avian plumage color gamut. Behavioural Ecology arr088. VIEW
Stoddard, M. C., & Stevens, M. 2010. Pattern mimicry of host eggs by the common cuckoo, as seen through a bird’s eye. Proceedings of the Royal Societ of London B: Biological Sciences rspb20092018. VIEW
Image credit
Top right: Audubon’s Warbler (Setophaga auduboni) © Pterzian CC BY SA 3.0 Wikimedia Commons.
If you want to write about your research in #theBOUblog, then please see here.




