LINKED PAPER
Breeding status influences timing but not duration of moult in the Northern Fulmar Fulmarus glacialis. Grissot, A., Graham, I.M., Quinn, L.,Bråthen, V.S. & Thompson, P.M. IBIS. DOI: 10.1111/ibi.12714. VIEW
I have always been interested in seabirds and their ecology, but never thought I’d have the chance to study them myself. Whilst studying for my Masters in France, an internship at the University of Aberdeen’s Lighthouse Field Station gave me the opportunity to carry out a project on Northern Fulmar (Fulmarus glacialis) moult phenology and lead to the publication of my first paper.
As an Early Career Researcher, writing your fist paper is clearly a milestone and can be very challenging. But it helps to write it on subject that fascinates you, and to develop the project in a team that helps you learn what it means to be a researcher. For me, it started when I was looking for a research project to complete my Master’s studies in Behavioural and Evolutionary Ecology: I had narrowed down my search to labs dealing with seabird ecology, and was lucky enough to be accepted at the Lighthouse Field Station in Cromarty, Scotland. The lab is working on marine species (Harbour Seals, Bottlenose Dolphins and Northern Fulmars) and this gave me the possibility to develop and lead a project on moult phenology of the Northern Fulmar. This project took me through every step of the research process, from collecting data in the field, to analysing it and obtaining results interesting enough to be presented in conferences and published in IBIS.
The Northern Fulmar is a seabird species from the Procellarid family, found primarily in subarctic regions of the North Atlantic and North Pacific oceans, where it is abundant and breeds in large colonies. Individuals leave the colony during the non-breeding season, and can be found over a broad wintering range. Many colonies are studied all over both the North Atlantic and North Pacific, and more and more knowledge is available about the ecology of the species. As a key marine species of the North Hemisphere, it is often used as an indicator of the environmental quality of the oceans, especially concerning plastic pollution (Van Franeker et al, 2011). Even though a lot is known about their breeding behaviour and phenology, these birds are harder to study during their non-breeding period because of their pelagic habits. Fortunately, current development of new technologies such as tracking devices can be of great help to know more about their whereabouts when away from the colony. Moreover, behaviour and phenology can now also be inferred from data gathered by these devices, as I had the chance to discover.
Figure 1 The location of the island of Eynhallow, Orkney, where the study took place, and view of the University of Aberdeen’s Lighthouse Field Station
The population studied during my placement, and on which is based the linked paper, is a perfect illustration of how a study can evolve as knowledge increases, new techniques become available and objectives change. The monitoring of this particular population started in the 1950’s with the idea to test a then popular hypothesis, that adult Fulmars do not breed every year (Wynne-Edwards, 1939; Dunnet, 1991). To test such a hypothesis, the catching and ringing of breeding adults started on the tiny Island of Eynhallow, Orkney (59°80N, 3°80W; Figure 1), and has been consistent since then. Numerous questions have since been raised and answered, taking inspiration on contemporary trends in Ecology and on development of new technologies and practices. That’s how Geolocation by light level (GLS) and GPS tracking have recently been introduced to the long-term monitoring; highlighting, for example, that individuals breeding at this colony can undertake journeys of over 6000km in 15 days to forage in the rich seas above the mid-ocean ridge (Edwards et al, 2013). The work I was entrusted to perform for the Lighthouse Field station has been one of the latest steps in this constantly changing process of research: using GLS tracking devices not only to look at movement and whereabouts of Fulmars, but also to obtain knowledge on their behaviour and phenology at the times when they are not directly observable.
 Figure 2 GLS device fixed on a darvic leg band
Figure 2 GLS device fixed on a darvic leg band
Most GLS devices now include other sensors in addition to the basic light sensor required to determine twice daily locations. The devices we used included a salt-water immersion logger that recorded immersion every 3s and stored the number of “wet” samples in a 5- or 10- minute period (depending on the brand and model used). As the logger is very small and fixed on a ring attached to a bird’s leg (Figure 2), it allowed us to know whether a bird was wet or dry at any 5- or 10- minute interval during the deployment, and determine its behaviour. The next step was to use the patterns observed in daily variation in behaviour (e.g. time spent sitting on the water) as a proxy for the timing of moult. This was based on two previous papers linking moult and a quasi-flightless period identifiable via salt water immersion data (Gutowsky et al. 2014; Cherel et al. 2016). Individual variation in the estimated timing of moult was then explained by life-history traits acquired through annual monitoring and individual based record of breeding history.
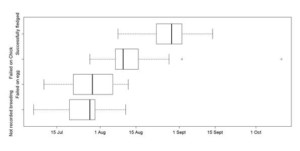
Figure 3 Differences in the starting date of the moult explained by breeding status. The boxes depict interquartile range, with median as a bold line. Whiskers indicate variability outside the upper and lower quartiles. Outliers are plotted as circles
We found a sex difference in the location of the moult, but not in its timing. However, the timing of moult did differ between individuals that had succeeded or failed in their previous breeding attempt, with successful breeders moulting the latest (Figure 3). In contrast, the duration of moult did not depend on prior reproductive success, but there was evidence of inter-annual variation in moult duration. This work highlights how GLS studies can provide a new step change in our understanding of key phases of the annual cycle, adding behavioural and phenological insight to our understanding of the at-sea distribution of pelagic seabirds.
Nominate this article for a BOU Science Communication Award.
References
Cherel, Y., Quillfeldt, P., Delord, K. & Weimerskirch, H. 2016. Combination of at-sea activity, geolocation and feather stable isotopes documents where and when seabirds molt. Front. Ecol. Evol. 4: 3. VIEW
Dunnet, G.M. 1991. Population studies of the Fulmar on Eynhallow, Orkney Islands. IBIS 133: 24–27. VIEW
Edwards, E.W.J., Quinn, L.R., Wakefield, E.D., Miller, P.I. & Thompson, P.M. 2013. Tracking a northern fulmar from a Scottish nesting site to the Charlie-Gibbs fracture zone: evidence of linkage between coastal breeding seabirds and Mid-Atlantic ridge feeding sites. Deep Sea Research Part II 98: 438–444. VIEW
Gutowsky, S.E., Gutowsky, L.F.G., Jonsen, I.D., Leonard, M.L., Naughton, M.B., Romano, M.D. & Shaffer, S.A. 2014. Daily activity budgets reveal a quasi-flightless stage during non-breeding in Hawaiian albatrosses. Mov. Ecol. 2: 23. VIEW
Van Franeker, J.A., Blaize, C., Danielsen, J., Fairclough, K., Gollan, J., Guse, N., Hansen, P.L., Heubeck, M., Jensen, J.K., Le Guillou, G., Olsen, B., Olsen, K.O., Pedersen, J., Stienen, E.W.M. & Turner, D.M. 2011. Monitoring plastic ingestion by the Northern Fulmar Fulmarus glacialis in the North Sea. Environ. Pollut. 159: 2609–2615. VIEW
Wynne-Edwards, V.C. 1939. Intermittent breeding of the Fulmar Fulmarus glacialis (L) with some general observations on nonbreeding in seabirds. Proc. 2001. SOC. Lond. (A) 109: 127-132. VIEW
Image credits
Featured image: Northern Fulmar, Fulmarus glacialis © Antoine Grissot




