LINKED PAPER
Patterns of association at feeder stations for Common Pheasants released into the wild: sexual segregation by space and time. Whiteside, M. A., van Horik, J. O., Langley, E. J. G., Beardsworth, C. E., Capstick, L. A. & Madden, J. R. 2019. IBIS. DOI: 10.1111/ibi.12632. VIEW
When you go birdwatching in spring, you will mostly encounter birds in pairs: a male and a female busily preparing for the breeding season. During the winter months, however, some birds tend to form sex-specific groups (Puttick 1981). This behaviour – called sexual segregation – has been thoroughly described in Pheasants (Phasianus colchicus). Males and females generally avoid each other until early March when groups of females start visiting the territories of males and the segregation breaks down (Hill & Ridley 1987).
This pattern can be explained by four factors. First, males and females might prefer different habitats. In China, for example, males roam the bamboo forests while females can be found in grasslands (Tso-Hsin 1964). Second, segregation might be due to different time budgets. In other words, males and females are active at different times of the day. Third, the sexes might differ in their dispersal patterns. Female pheasants often disperse further and more quickly compared to males (Hill & Ridley 1987). Finally, some individuals may simply prefer to hang out with members of the same sex. To discriminate between these four factors, ornithologists from the University of Exeter performed a large-scale experiment in Devon, UK. They released 200 captive-reared Pheasants and tracked their movements during the following months.
Time and space
The researchers monitored the Pheasants at 40 feeding stations. Each feeder was accessible for one bird at a time. However, the Pheasants would arrive in clusters and take turns at the feeder or just wander around without feeding. Because each bird was individually marked with wing tags, the researchers could easily determine which birds enjoyed each other’s company. The analyses of over 30,000 visits revealed some interesting patterns. First, there was spatial segregation: males mostly visited feeders in the south of the study area, whereas females tended to use feeders in the north. This pattern could point to different habitat preferences between the sexes, but the environment in the study area was too homogeneous to draw this conclusion.
Second, there was temporal segregation: females were more likely to visit feeders in the morning, while males showed up in the afternoon. This result could be the outcome of sex-specific time schedules. Males might spend the morning hidden in the forest, recovering from their territorial defense during dusk and dawn (Heinz & Gysel 1970). Or perhaps predation risk varies during the day for males and females? But this hypothesis requires a more detailed understanding of the predators in the area.
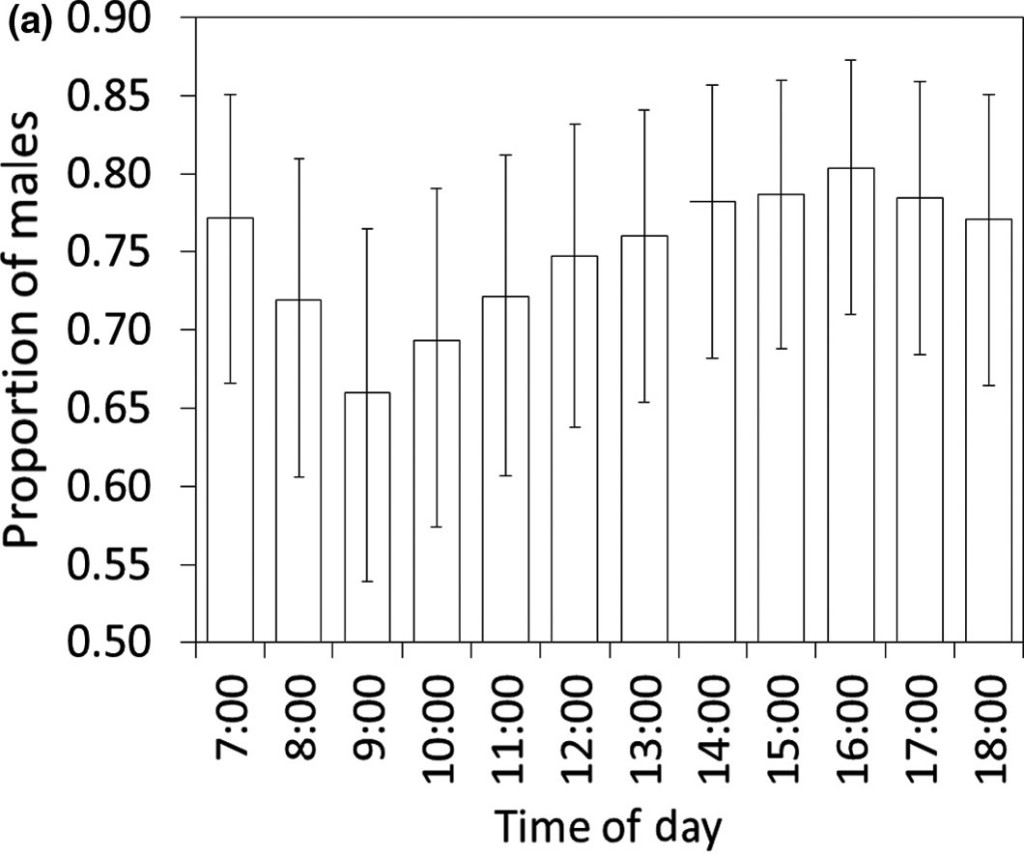
Figure 1 The proportion of males visiting the feeders at different times of the day shows a clear decrease in the morning. Could the males be recovering from their territorial defense?
Social network
Finally, the analyses of the social interactions showed that the social networks changed over time. At the beginning of the experiment (in August), males tended to associate more with other males, while females did not aggregate. As the experiment progressed, this pattern reversed. By November, females showed clear signs of aggregation, whereas males tended to avoid other males. The avoidance behaviour of males points to the establishment of territories. However, some males continued to from distinct groups. These birds probably took longer to establish a territory or they might be satellite males that did not hold territories during the breeding season (Hill & Robertson 1988).
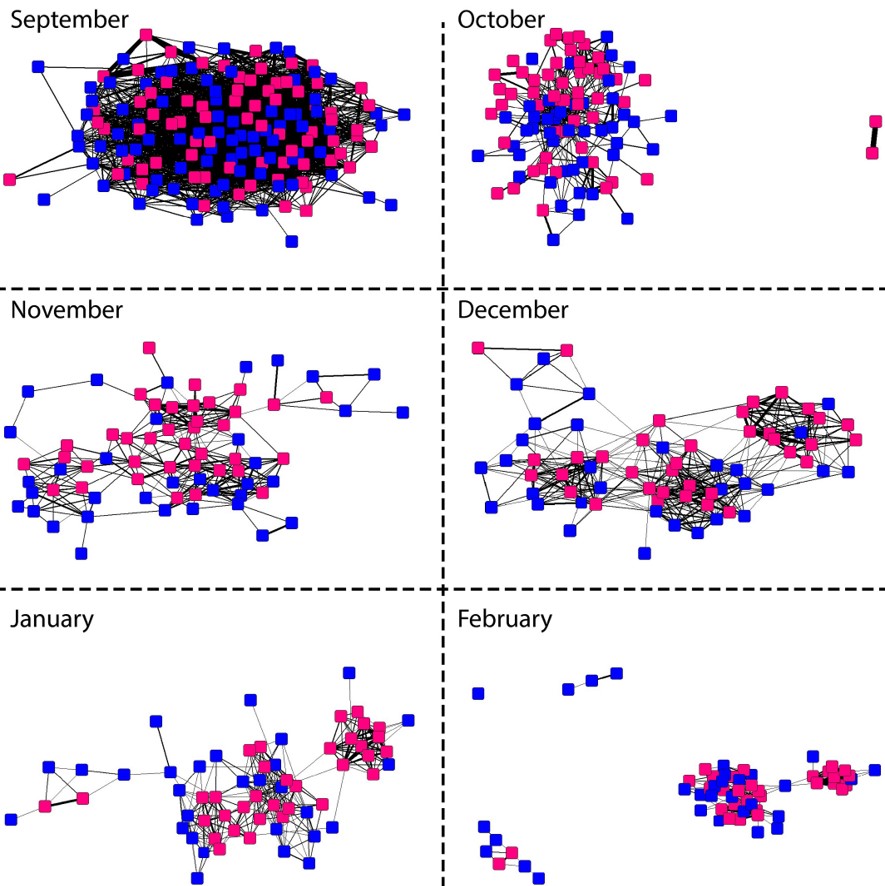 Figure 2 The social network of Pheasant interactions changed during the experiment. In the beginning, males (blue) tended to aggregate while females (pink) avoided contact. Later on, females started to form distinct clusters.
Figure 2 The social network of Pheasant interactions changed during the experiment. In the beginning, males (blue) tended to aggregate while females (pink) avoided contact. Later on, females started to form distinct clusters.
Unnatural
This experiment nicely shows how sexual segregation in Pheasants plays out in time and space. However, the researchers are aware that the observations were obtained in a somewhat unnatural environment. The birds were captive-reared and quickly learned the locations of feeding stations. Whether these patterns of segregation hold in wild populations remains to be investigated (but see Hill & Ridley 1987). Nonetheless, the combination of experiments and field studies provides an exciting avenue for further research into Pheasant behaviour.
References
Heinz, G. H. & Gysel, L. W. 1970. Vocalization Behavior of the Ring-Necked Pheasant. The Auk 87: 279-295. VIEW
Hill, D. A. & Ridley, M. W. 1987. Sexual segregation in winter, spring dispersal and habitat use in the pheasant (Phasianus colchicus). Journal of Zoology 212: 657-668. VIEW
Hill, D. A. & Robertson, P. A. 1988. The Pheasant: Ecology, Management and Conservation. Oxford: Blackwell Scientific Books. VIEW
Puttick, G. M. 1981. Sex-related differences in foraging behaviour of Curlew Sandpipers. Ornis Scandinavica 12: 13-17. VIEW
Tso-Hsin, C. 1964. China’s economic fauna: Birds. Washington DC: US Department of Commerce’s Office of Technical Service, Joint Publication Research Service. VIEW
Image credits
Featured image: Pheasant Phasianus colchicus | Lukasz Lukasik | CC BY-SA 3.0 Wikimedia Commons




