Chick predation CSI in a high-stakes game of predator prey
LINKED PAPER
Tracking day and night provides insights into the relative importance of different wader chick predators. Mason, L.R., Smart, J. & Drewitt, A.L. 2017. IBIS. DOI: 10.1111/ibi.12523. VIEW
Picture this. It’s a glorious summer day in the Norfolk Broads, and you’re surrounded by nothing but a huge expanse of grazing marsh and the calls of lowland waders. You’re carrying a radio receiver and antenna, from which comes the slow incessant beeping of a radio tag that you attached to a Lapwing chick, Vanellus vanellus, just days ago.
Beep, beep, beep…

Figure 1 Lapwing chick ringed, flagged and tagged ready for action © Ros Kennerley
Today there’s no sign of that chick or its siblings in the field, but, if you stand on the back of your LandRover you can hear the tag signal more strongly: the chick must be a long way away. As you follow the direction of the tag signal and it gets steadily louder, you realise you’re now 1km away from where the chick hatched. Definitely not a good sign! Your heart sinks.
Beep, beep, beep…
Finally, your ears take you a large hole in a bank. A fox earth. The tag is lying encased in a fox scat outside it, still beeping. Further searching in the area reveals the remains of chicks from other broods outside and inside the earth. At least five of your carefully monitored chicks have met their grisly end in the stomach of a fox family.

That is a true story from my summer in 2010, which I spent as a detective in a high-stakes game of predator and prey; and the detective work did not end there. I monitored 46 lapwing chicks that year, and my job was to determine the type or species of predator involved in the deaths of 42 of them. One other notable find being in a kestrel nest in a tree on the other side of an estuary 2km away (the first time I’ve ever had to use a boat and a ladder to find a lapwing chick)!
All this detective work was for a reason: wader populations are in trouble in the UK and across western Europe, with many species unable to produce enough young to counter adult mortality because of low nest and chick survival (Roodbergen et al. 2012). Predation is a key reason for this, particularly now that many waders are restricted to increasingly-isolated pockets of suitably-wet habitat, which attract predators like honeypots attract bees.
Before you can manage the effects of predation you first need to understand who the predators are. But how do you do this for wandering wader chicks? The traditional way is to become a detective by fitting chicks with radio tags, monitoring them in case they are predated, then tracking down their predated bodies in the hope that you a) can find them, and b) can get some clue about the predator from the state and location of the remains.
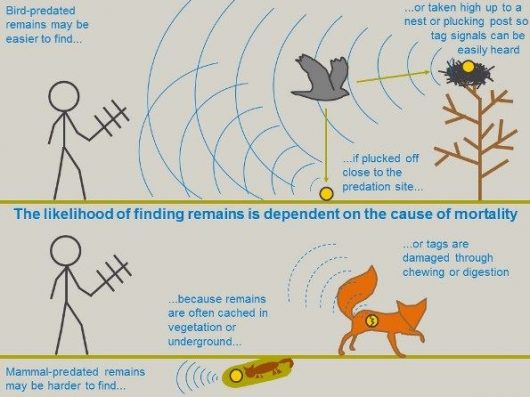
But manual tracking is inherently flawed when deducing “whodunit”. Rarely do you find the remains of every chick, and you are more likely to find remains predated by certain predator groups. To remove some of this uncertainty, we decided to determine the timing of chick predation using Automatic Radio Tracking Stations (ARTS), which recorded signals from radio-tagged lapwing chicks around the clock and pinpointed the time of predation to within 10 minutes, thus incriminating diurnal or nocturnal predators. This was valuable because on lowland wet grassland the predators can be grouped into those active at night (mammals) and those active during the day (primarily birds, but also mammals that hunt by day).
Between 2009 and 2013, we monitored 179 lapwing chicks on 15 lowland wet grassland sites in the UK, combining ARTS tracking with traditional manual tracking and rapid monitoring of the predators themselves (faecal/scat transects for foxes, tracking tunnels for mustelids, and timed watches for avian predators) to provide insights into the relative importance of different wader chick predators.

The first surprise was the level of predation we encountered. 87% of our chicks were predated: a shockingly high proportion considering those were the chicks that managed to survive nest predation in the first place.
Also surprisingly, although day-time predation was the most common, it was nocturnal predation that was more intensive, with chicks more likely to be predated per hour at night. Mammalian predation was also more important than avian predation, reflecting the high nocturnal predation rate and also the partial contribution of mammals to predation during the day.
And, as my detective story hinted at, foxes were the principle culprits of mammalian predation and were the only predator group that statistically influenced chick predation probability. Where fox activity was high, the probability that a chick would survive 30 days in the field was < 2%.

So what do we do about this? We found that chick predation was less likely at the start of the breeding season than at the end, meaning that if we can encourage successful early nesting attempts then early-hatched chicks are more likely to fledge.
We also found that the proportion of chicks predated by three main predator groups (foxes, small mustelids, raptors) were directly related to the activity of those predators on a site, so predator activity monitoring could indicate which of these groups will impact the most. Another positive note is that the chicks we monitored were in otherwise good condition, so could be expected to fledge successfully if we can reduce predation rates on well-managed wet grassland sites (Mason & Smart 2015).
The next steps to develop more sustainable predation management methods are already underway. Research published by Rebecca Laidlaw has shown that nest survival can be improved by providing long grass areas outside wader fields to attract small mammals and encourage predators to concentrate on hunting these instead (Laidlaw et al. 2016). This method could be combined with other predation management methods such as electric fencing, diversionary feeding and lethal control on and off reserves to manage predation going forward (Bolton et al. 2007; Malpas et al. 2013).
A huge thanks to all the wader detectives who took part in this project, and all the sites that made it possible.
References and further reading
Appleton, G. 2015. How well do Lapwings and Redshanks grow? Wader Tales, November 30, 2015. Available from: https://wadertales.wordpress.com/2015/11/30/how-well-do-lapwings-and-redshanks-grow/. VIEW
Appleton, G. 2017a. Can habitat management rescue Lapwing populations? Wader Tales, January 5, 2017. Available from: https://wadertales.wordpress.com/2017/01/05/can-habitat-management-rescue-lapwing-populations/. VIEW
Appleton, G. 2017b. Mastering Lapwing conservation. Wader Tales, June 7, 2017. Available from: https://wadertales.wordpress.com/2017/06/07/mastering-lapwing-conservation/. VIEW
Bolton, M., G. Tyler, K. Smith, and R. Bamford. 2007. The impact of predator control on lapwing Vanellus vanellus breeding success on wet grassland nature reserves. Journal of Applied Ecology 44: 534-544. VIEW
Laidlaw, R.A., Smart, J., Smart, M.A. & Gill, J.A.. 2016. Scenarios of habitat management options to reduce predator impacts on nesting waders. Journal of Applied Ecology 54: 1219-1229. VIEW
Malpas, L.R., Kennerley, R.J., Hirons, G.J.M., Sheldon, R.D., Ausden, M., Gilbert, J.C. & Smart, J. 2013. The use of predator-exclusion fencing as a management tool improves the breeding success of waders on lowland wet grassland. Journal for Nature Conservation 21: 37-47. VIEW
Mason, L.R. & Smart, J. 2015. Wader chick condition is not limited by resource availability on wader-friendly lowland wet grassland sites in the UK. Wader Study 122: 193-200. VIEW
Roodbergen, M., van der Werf, B. & Hötker, H.. 2012. Revealing the contributions of reproduction and survival to the Europe-wide decline in meadow birds: review and meta-analysis. Journal of Ornithology 153: 53-74. VIEW
Teunissen, W., Schekkerman, H., Willems, F. & Majoor, F. 2008. Identifying predators of eggs and chicks of Lapwing Vanellus vanellus and Black-tailed Godwit Limosa limosa in the Netherlands and the importance of predation on wader reproductive output. IBIS 150: 74-85. VIEW
If you want to write about your research in #theBOUblog, then please see here.





